Gene Therapy
Lipofection of nondividing cells is inefficient because much of the transfected DNA is retained in endosomes, and that which escapes to the cytoplasm enters the nucleus at low rates. To improve the final ratelimiting step of nuclear import, we have used the nonclassical nuclear localization signal (NLS) containing the M9 sequence of heterogeneous nuclear ribonucleoprotein (hnRNP) A1. This approach has been validated in endothelial cells, neurons, and mouse embryonic stem cells. 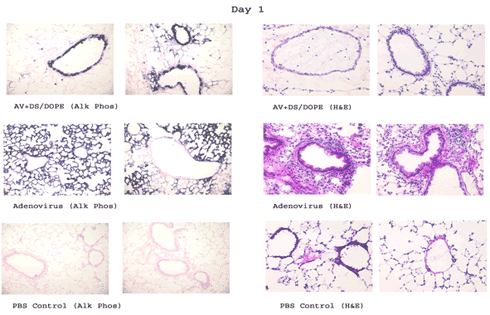
During the delivery of plasmid DNA for gene therapy often provokes an inflammatory response that reduces transgene expression. Cationic lipids for lipofection lack pharmacological activity despite the hydrophobicity of many drug candidates that could be exploited. We have developed a one-step synthesis of a water-soluble, dexamethasone–spermine (DS) cationic lipid that has potent gene transfer capability in confluent endothelial cells when used with the neutral lipid, dioleoylphosphatidylethanolamine (DOPE). DS retains partial corticosteroid character as quantified by the rapid translocation of glucocorticoid receptor to the nucleus and by dosedependent transactivation from a glucocorticoid response element. DS has anti-inflammatory activity in vivo in the mouse thioglycollate model of inflammation. In a mouse lung model, DS:DOPE resulted in significantly less interferon-g production at Day 1 and elevated transgene expression at Days 1 and 7 postintranasal instillation compared to DCChol: DOPE.
We formulated adenovirus (AdV) vectors with cationic steroid liposomes containing dexamethasone–spermine (DS)/dioleoylphosphatidylethanolamine (DOPE) in an effort to overcome the lack of apically expressed AdV vector receptors on airway epithelial cells and to reduce the inflammation associated with AdV vector exposure. An AdV vector (1 to 2.5 x10^11 genome copies) expressing human placental alkaline phosphatase or B-galactosidase (LacZ) was delivered alone or complexed with DS/DOPE, DC-Chol/DOPE, or dexamethasone to C57Bl/6 mice via intranasal instillation. Formulation of the AdV vector with DS/DOPE and DC-Chol/DOPE resulted in transgene expression targeted only to the airway epithelial cells with minimal expression in alveolar cells, while AdV alone caused high alveolar transduction. The DS/DOPE and dexamethasone formulations greatly reduced cellular infiltrates compared to AdV vector alone, while formulation with DC-Chol/DOPE did not. Viral vectors can be formulated with DS/DOPE to improve targeting to the airway epithelium in vivo and to attenuate vector-induced inflammation through the pharmacological activity of DS.
Publications
| 1. |
1997 |
Subramanian A, Diamond SL. Enhancement of nonviral gene transfer to endothelial cells using lipofection of histone-complexed DNA. Tissue Engineering 1997; 3 (1): 39-52  |
| 2. |
1999 |
Subramanian A, Ranganathan P, Diamond SL. Nuclear targeting peptide scaffolds for lipofection of nondividing mammalian cells. Nat Biotechnol 1999; 17 (9): 873-877  |
| 3. |
2001 |
Ma H, Diamond SL. Nonviral gene therapy and its delivery systems. Curr Pharm Biotechnol 2001; 2 (1): 1-17  |
| 4. |
2002 |
Byrnes CK, Nass PH, Duncan MD, Diamond SL, Harmon JW. Assessment of a Nuclear Targeting Peptide Scaffold (M9) to Improve Plasmid DNA Transfection Efficiency. Surgical Forum 2002; 52 : 538-539  |
| 5. |
|
Ma H, Zhu J, Maronski M, Kotzbauer PT, Lee VM, Dichter MA, Diamond SL. Non-classical nuclear localization signal peptides for high efficiency lipofection of primary neurons and neuronal cell lines. Neuroscience 2002; 112 (1): 1-5  |
| 6. |
|
Subramanian A, Ma H, Dahl KN, Zhu J, Diamond SL. Adenovirus or HA-2 fusogenic peptide-assisted lipofection increases cytoplasmic levels of plasmid in nondividing endothelium with little enhancement of transgene expression. J Gene Med 2002; 4 (1): 75-83  |
| 7. |
2003 |
Pierce EA, Liu Q, Igoucheva O, Omarrudin R, Ma H, Diamond SL, Yoon K. Oligonucleotide-directed single-base DNA alterations in mouse embryonic stem cells. Gene Ther 2003; 10 (1): 24-33  |
| 8. |
2004 |
Gruneich JA, Price A, Zhu J, Diamond SL. Cationic corticosteroid for nonviral gene delivery. Gene Ther 2004; 11 (8): 668-674  |
| 9. |
2005 |
Price A, Limberis M, Gruneich JA, Wilson JM, Diamond SL. Targeting viral-mediated transduction to the lung airway epithelium with the anti-inflammatory cationic lipid dexamethasone-spermine. Mol Ther 2005; 12 (3): 502-509  |
| 10. |
2006 |
Kim MS, Diamond SL. Photocleavage of o-nitrobenzyl ether derivatives for rapid biomedical release applications. Bioorg Med Chem Lett 2006; 16 (15): 4007-4010  |
| 11. |
|
Kim MS, Diamond SL. Controlled release of DNA/polyamine complex by photoirradiation of a solid phase presenting o-nitrobenzyl ether tethered spermine or polyethyleneimine. Bioorg Med Chem Lett 2006; 16 (21): 5572-5575  |
| 12. |
2007 |
Gruneich JA, Diamond SL. Synthesis and structure-activity relationships of a series of increasingly hydrophobic cationic steroid lipofection reagents. J Gene Med 2007; 9 (5): 381-391  |
| 13. |
|
Handwerger RG, Diamond SL. Biotinylated photocleavable polyethylenimine: capture and triggered release of nucleic acids from solid supports. Bioconjug Chem 2007; 18 (3): 717-723  |
| 14. |
|
Murphy BR, Moayedpardazi HS, Gewirtz AM, Diamond SL, Pierce EA. Delivery and mechanistic considerations for the production of knock-in mice by single-stranded oligonucleotide gene targeting. Gene Ther 2007; 14 (4): 304-315  |
| 15. |
|
Price AR, Limberis MP, Wilson JM, Diamond SL. Pulmonary delivery of adenovirus vector formulated with dexamethasone-spermine facilitates homologous vector re-administration. Gene Ther 2007; 14 (22): 1594-1604  |
| 16. |
2008 |
Barker GA, Diamond SL. RNA interference screen to identify pathways that enhance or reduce nonviral gene transfer during lipofection. Molecular Therapy 2008; 16 (9): 1602-1608  |
| 17. |
2009 |
Fein DE, Limberis MP, Maloney SF, Heath JM, Wilson JM, Diamond SL. Cationic lipid formulations alter the in vivo tropism of AAV2/9 vector in lung. Mol Ther 2009; 17 (12): 2078-2087  |
| 18. |
2010 |
Fein DE, Bucki R, Byfield F, Leszczynska K, Janmey PA, Diamond SL. Novel cationic lipids with enhanced gene delivery and antimicrobial activity. Mol Pharmacol 2010; 78 (3): 402-410  |
| 19. |
2011 |
Wallen AJ, Barker GA, Fein DE, Jing H, Diamond SL. Enhancers of adeno-associated virus AAV2 transduction via high throughput siRNA screening. Mol Ther 2011; 19 (6): 1152-1160  |
|





